Bioburden Testing Market Outlook: A Strategic Roadmap to 2031
The pharmaceutical and medical technology sectors are currently operating under some of the most rigorous safety mandates in history. At the center of this transition is a renewed focus on microbial control and sterilization validation. Understanding the Bioburden Testing Market Outlook is essential for manufacturers aiming to navigate the complexities of product safety, regulatory compliance, and rapid production cycles.
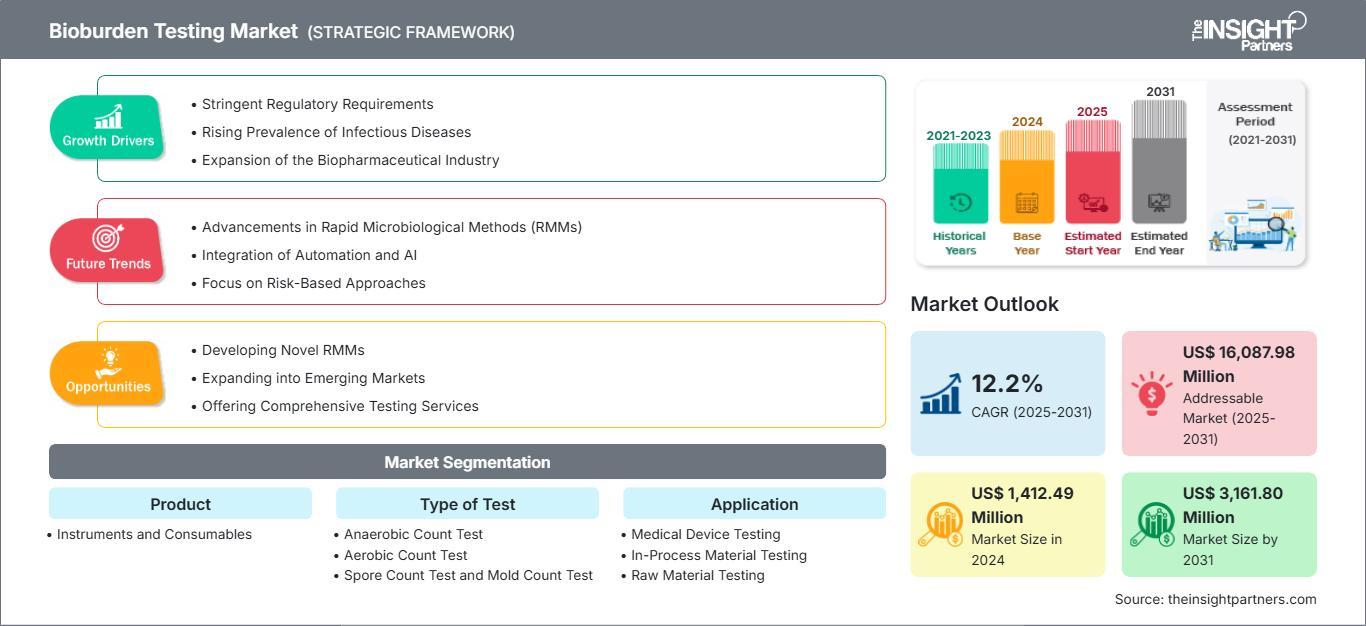
The Bioburden Testing Market size is expected to reach US$ 3,162 million by 2031. The market is anticipated to register a CAGR of 12.2% during 2025–2031. This growth is a direct reflection of the rising standards for sterile manufacturing and the increasing volume of complex biologics entering the pipeline.
Download Sample Report - https://www.theinsightpartners.com/sample/TIPRE00003000
Critical Market Drivers Shaping the Outlook
The current Bioburden Testing Market Outlook is defined by several transformative drivers that are shifting how laboratories and manufacturers approach microbial enumeration:
Heightened Regulatory Scrutiny The most significant driver is the tightening of global regulatory frameworks. Compliance with ISO 11737 and updated Annex 1 guidelines in Europe has made bioburden testing a mandatory, non negotiable step in the product lifecycle. Regulatory agencies now demand more frequent testing and more granular data on microbial populations to ensure that sterilization cycles are not only effective but also optimized for the specific microbial load of each batch.
Rise in Biopharmaceutical Complexity The industry is moving away from simple small molecule drugs toward complex biologics, vaccines, and cell therapies. These products are highly susceptible to contamination during the "in process" stage. Unlike traditional medical devices, biologics require highly sensitive bioburden testing to detect low level microbial presence that could otherwise compromise the entire batch. This specialized need is driving the demand for advanced reagents and high precision instruments.
Surge in Medical Device Recalls Recent years have seen an uptick in product recalls due to sterility failures. To protect brand reputation and patient safety, manufacturers are adopting proactive bioburden monitoring strategies. By identifying potential contamination at the raw material or in process stage, companies can avoid the catastrophic costs associated with post market recalls. This shift from reactive to proactive testing is a primary catalyst for market expansion.
Top Players in the Bioburden Testing Market
The market is led by a group of innovative companies providing both cutting edge hardware and the essential consumables required for routine testing. These top players include:
- Thermo Fisher Scientific Inc.
- Merck KGaA
- bioMérieux SA
- BD (Becton, Dickinson and Company)
- QIAGEN
- SGS SA
- Pacific BioLabs
- Nelson Laboratories, LLC
- Charles River Laboratories
- STERIS
Technological Innovations: The Future of Testing
One of the most exciting aspects of the Bioburden Testing Market Outlook is the integration of Digital Microbiology. Traditional plate count methods, which often take days to produce results, are being supplemented by Rapid Microbiological Methods (RMMs). Technologies such as ATP bioluminescence and automated colony counters are reducing results turnaround from days to hours.
Furthermore, the adoption of Artificial Intelligence (AI) for image analysis in colony counting is significantly reducing human error. These digital tools allow for better data traceability, making it easier for manufacturers to provide clear audit trails during government inspections.
Regional Growth and Outsourcing Trends
While North America remains the largest hub for bioburden testing due to its concentrated pharmaceutical sector, the Asia Pacific region is rapidly catching up. The growth in APAC is fueled by the expansion of Contract Manufacturing Organizations (CMOs). Many global firms are outsourcing their testing requirements to specialized labs in this region to leverage cost efficiencies and specialized expertise. This trend is expected to continue as the Bioburden Testing Market Outlook remains positive for emerging economies.
Conclusion
As we look toward 2031, the Bioburden Testing Market Outlook remains strong, supported by a CAGR of 12.2% and a clear shift toward automated, rapid testing solutions. For manufacturers, the path forward involves investing in these advanced technologies to ensure that product safety never comes at the cost of production speed.
Related Report :
· Pharmaceutical Quality Control Market Overview, Growth, Trends, Research Report (2021-2031)
Contact Information -
Email: sales@theinsightpartners.com
Phone: +1-646-491-9876
Also Available in :
Korean German Japanese French Chinese Italian Spanish




